Scientists See Electron Bottleneck in Simulated Battery
Molecular Foundry researchers help shed light on barrier to optimal performance.

The Science
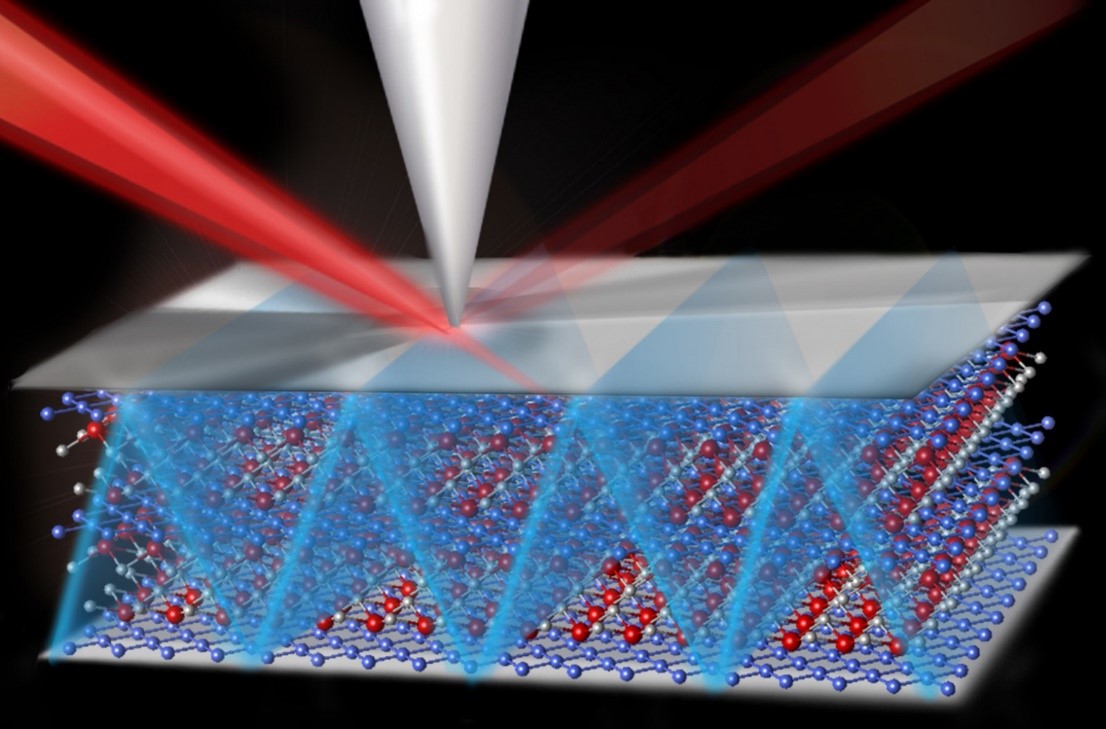
As the appetite grows for more efficient vehicles and mobile devices based on cleaner, renewable energy sources, so does the demand for batteries that pack more punch, last longer, and charge or discharge more quickly. The compound vanadium pentoxide has grabbed the spotlight as a way to improve lithium-ion batteries. However, it’s less-than-stellar behavior has been problematic. An international team working at the Molecular Foundry revealed why the material may not perform as expected. The team discovered how interactions between electrons and ions slow the performance of electrodes made with vanadium pentoxide.
The Impact
This work answers, in part, why the material gets bogged down. Vanadium pentoxide’s layered atomic structure results in a vast surface area, but a bottleneck occurs. If scientists can address the bottleneck, this material may lead to the next generation of batteries, which pack more punch, last longer, and charge or discharge more quickly.
Summary
An international team of scientists working at the Molecular Foundry has revealed how interactions between electrons and ions can slow down the performance of vanadium pentoxide, a material considered key to the next generation of batteries. The compound vanadium pentoxide has grabbed the spotlight as a potential nanostructured material for state-of-the-art lithium-ion batteries because it can provide a greater surface area for the arrival and insertion of lithium ions. That quality makes vanadium pentoxide a good candidate as a cathode, the part of a battery where electrons and lithium ions enter.
The speed with which electrons can enter and exit the cathode determines how much power the battery can provide. The entry and exit speed also determine how quickly a battery recharges. Power density and charging are both critical factors in the world of mobile electronics or electrification of our automotive fleet. But despite vanadium pentoxide’s potential, it has yet to be widely adopted commercially because of its less-than-stellar performance when put to the test in the real world.
The new findings shed light on the slowdown. The results show that the flow of electrons in vanadium pentoxide nanowires gets bogged down as it interacts with lithium ions in a phenomenon known as small polaron formation. The research group, which involved scientists at Texas A&M University, made 2D maps of the electronic properties of synthesized vanadium pentoxide nanowires serving as a model lithium-ion cathode using scanning transmission x-ray microscopy at the Canadian Light Source. They came to the Molecular Foundry to interpret their findings.
Contact
David Prendergast
Molecular Foundry, Berkeley Lab
dgprendergast@lbl.gov; 510.486.4948
Funding
This study is based on work supported by the National Science Foundation under DMR 1504702. S.B. further acknowledges support from the Research Corporation for Science Advancement through a Scialog Award. L.D.J. acknowledges support from a National Science Foundation Graduate Research Fellowship under grant 1252521. The scientists acknowledge Chithra Karunakaran at beam-line 10ID1 of the Canadian Light Source for support and assistance with scanning transmission x-ray microscopy (STXM) data collection. Use of the National Synchrotron Light Source, Brookhaven National Laboratory, was supported by the U.S. Department of Energy, Office of Science, Office of Basic Energy Sciences, under contract DE-AC02-98CH10886. Density functional theory (DFT) simulations were performed as part of a user project with Y.L. and D.P. at the Molecular Foundry (TMF), Lawrence Berkeley National Laboratory, and calculations were executed on their Vulcan and Nano compute clusters, administered by the High-Performance Computing Services Group at Lawrence Berkeley National Laboratory. TMF is supported by the Office of Science, Office of Basic Energy Sciences, of the U.S. Department of Energy, under contract DE-AC02-05CH11231. We thank Joseph Woicik for access and assistance at the X24a end station. Acknowledgement is made to the Donors of the American Chemical Society Petroleum Research Fund (PRF 52827-DNI10) for support of the research at Binghamton University.
Publications
L.R. De Jesus, G.A. Horrocks, Y. Liang, A. Parija, C. Jaye, L. Wangoh, J. Wang, D.A. Fischer, L.F.J. Piper, D. Prendergast, and S. Banerjee, “Mapping polaronic states and lithiation gradients in individual V2O5 nanowires.” Nature Communications 7, 12022 (2016). [DOI: 10.1038/ncomms12022]
Related Links
Lawrence Berkeley National Laboratory news release: Scientists See Electron Bottleneck in Simulated Battery
Highlight Categories
Performer: University , DOE Laboratory , SC User Facilities , BES User Facilities , NSLS , Foundry
Additional: Collaborations , Non-DOE Interagency Collaboration , International Collaboration