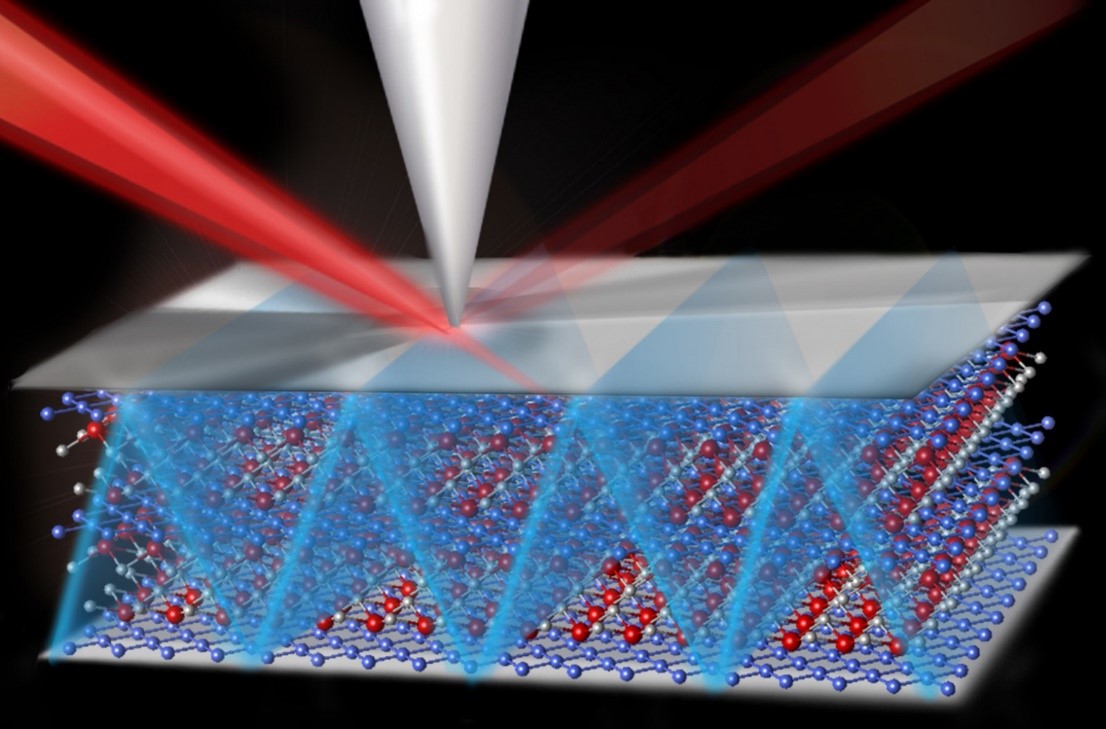
Adding Nanocavities to Catalyst Surfaces Enhances Chemical Selectivity
Adding an oxide sieve, a layer containing nanocavities, to a catalyst surface makes the catalyst selective for specific reactions and increases efficiencies for chemical processes.
The Science
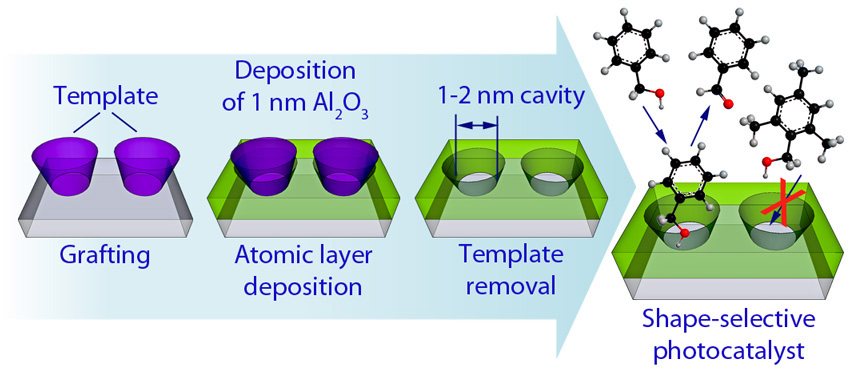
Using a molecular template before atomic layer deposition leads to cavities in thin oxide films that act as shape-selective sieving layers for oxidations and reductions.
The Impact
Solid oxide catalysts are widely used in emissions reduction, petrochemical and biomass conversions, and other reactions, but unlike zeolite catalysts, are generally not shape-selective. This new synthesis method can modify existing, highly active, catalysts to add selectivity. This will result in less-costly processes through decreased need for separations and greatly improved yields.
Summary
Catalysts are compounds that enhance the speed of chemical reactions in a wide range of industrial chemistry applications. The Institute for Atom-Efficient Chemical Transformation (IACT), a DOE-supported Energy Frontier Research Center (EFRC), is developing new reactions and new catalytic materials for bioenergy production by combining advanced characterization, computer simulations, and materials synthesis. IACT researchers at Northwestern University and Argonne National Laboratory developed a new technique to modify existing oxide catalysts with a surface film that enhances the selectivity of the catalyst. These films contain <2 nm diameter “nanocavities” made by adding a template during the atomic layer deposition process that is used to synthesize atom-precise films of oxides, metals, and other materials. Removal of the template after synthesis results in the nanocavities in the surface that provides a sieving effect, allowing separation of the reactant molecules and limiting reactions to a single particle. Because the thickness of the sieving layer is comparable in size to the reactant molecules, diffusional limitations that plague other materials are not present here. Ultimately, adding selectivity to intrinsically non-selective oxide catalysts is expected to decrease the cost for biofuels and bio-derived chemicals by decreasing the need for challenging and costly separations and increasing the product yields.
The research utilized the Advanced Photon Source for characterization of the structures.
Contact
Justin M Notestein
Northwestern University
j-notestein@northwestern.edu
Chris Marshall
Director of the Institute for Atom-Efficient Chemical Transformation EFRC
marshall@anl.gov
Funding
DOE Office of Science, Basic Energy Sciences (BES), Energy Frontier Research Centers (EFRC) Program. The Advanced Photon Source is an Office of Science User Facility supported by BES. JMN acknowledges a 3M Non-Tenured Faculty Grant, a DuPont Young Professor Grant, a Camille and Henry Dreyfus New Faculty Award, and U.S. Department of Energy award DE-SC0006718 for supplemental funding of materials characterization and supplies. TEM was performed at the Electron Probe Instrumentation Center of NUANCE at Northwestern University, supported by the National Science Foundation, the Keck Foundation, and the State of Illinois.
Publications
C. P. Canlas, J. Lu, N. A. Ray, N. A. Grosso-Giordano, J. W. Elam, S. Lee, R. E. Winans, P. C. Stair, R. P. Van Duyne, and J. M. Notestein, “Shape-Selective Sieving Layers on an Oxide Catalyst Surface,” Nature Chem., 2012, 4, 1030–1036. [DOI: 10.1038/nchem.1477]
Related Links
Northwestern Univeristy McCormick Magazine article
Chemical and Engineering News article
Institute for Atom-Efficient Chemical Transformation (IACT) EFRC
Highlight Categories
Performer: University , DOE Laboratory , SC User Facilities , BES User Facilities , APS