New Method Knocks Out Yeast Genes with Single-Point Precision
Researchers can precisely study how different genes affect key properties in a yeast used industrially to produce fuel and chemicals.
The Science
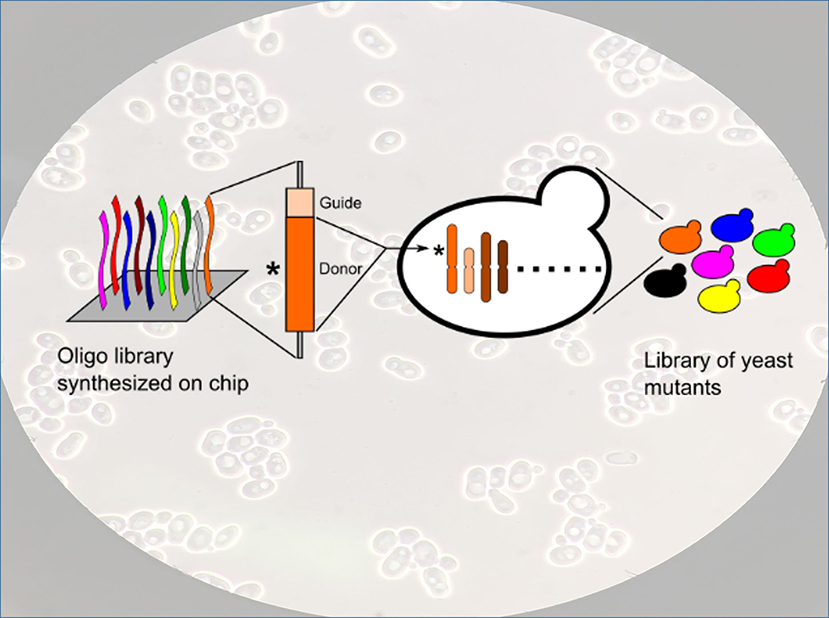
How do you make yeast work harder? Not to make bread, but in processes that yield chemicals and pharmaceuticals. Industries currently use a yeast called Saccharomyces cerevisiae. They’d like it to work better. The answer is in manipulating the yeast’s genetic code. To get at that code, researchers developed a method that turns off targeted genes in the yeast, introducing mutations. The team’s approach deletes specific points in the DNA sequence. They study how each deletion affects the yeast. Does a deletion cause the yeast to stop working in certain chemicals? Does a deletion make the yeast grow more slowly? The team’s approach lets them study each gene, as well as in combination with other genes. With this approach, scientists can construct libraries of mutants for use in discovering how each gene works.
The Impact
Libraries of genetic mutations have so far only been achieved in simpler organisms, specifically prokaryotes. Now, scientists can build such libraries for more complex organisms. The new technique lets scientists rapidly engineer tens of thousands of genes. They can target the genes with 98 percent efficiency. The results ease identifying and isolating mutant strains that show desired traits, such as tolerance to toxic compounds necessary to produce industrial products.
Summary
Researchers developed a method called CRISPRCas9- and homology-directed-repair-assisted genome-scale engineering (CHAnGE) using libraries of synthetic oligonucleotides (cassettes) containing a CRISPR guide sequence, gene-specific sequences to target homologous recombination to those selected genes, and unique barcodes to track each mutant strain. The oligonucleotide library was cloned into a plasmid and introduced into Saccharomyces cerevisiae. Nearly 25,000 sequences representing almost every one of the 6,500 yeast open reading frames were synthesized. More than 98 percent of the CHAnGE cassettes resulted in mutations in the target genes at least 82 percent of the time, demonstrating a high editing efficiency. The technology proved to be effective for the introduction of both small deletions and single-base mutations, as well as for saturation mutagenesis of a single gene or domain. CHAnGE was successfully applied to engineer yeast strains that are tolerant to furfural, indicating that it could be used to engineer industrially relevant eukaryotes to advance toward renewable production of biofuels and valuable chemicals.
Contact
Program Manager
Pablo Rabinowicz
Department of Energy, Office of Science, Biological and Environmental Research
pablo.rabinowicz@science.doe.gov
Principal Investigator
Huimin Zhao
University of Illinois at Urbana-Champaign
zhao5@illinois.edu
Funding
This work was supported by the Office of Biological and Environmental Research within the Department of Energy’s Office of Science and the Carl R. Woese Institute for Genomic Biology at the University of Illinois at Urbana-Champaign.
Publications
Z. Bao, M. HamediRad, P. Xue, H. Xiao, I. Tasan, R. Chao, J. Liang, and H. Zhao, “Genome-scale engineering of Saccharomyces cerevisiae with single-nucleotide precision.” Nature Biotechnology 36, 505 (2018). [DOI: 10.1038/nbt.4132]
Related Links
University of Illinois press release: New CRISPR technology ‘knocks out’ yeast genes with single-point precision
Highlight Categories
Performer: University
Additional: Collaborations , Non-DOE Interagency Collaboration